Radiotherapy for early Dupuytren’s disease can slow progression and preserve hand function, yet it is not routinely available on the NHS. This article explains how treatments are assessed by NICE, why evidence structure and funding pressures matter, and why most patients currently access this treatment privately despite supportive clinical data.
Radiotherapy for early Dupuytren’s disease is technically permitted in the UK, but it is not routinely commissioned or funded by the NHS. In practical terms, that means patients who wish to pursue it almost always do so privately. To understand why, it helps to look at how treatments become funded within the NHS.
How Treatments Are Evaluated
In the UK, new or emerging treatments are usually assessed by NICE – the National Institute for Health and Care Excellence.
NICE evaluates two things at the same time: the strength and structure of the clinical evidence, and whether a treatment represents value for money within a finite NHS budget. Those two considerations are inseparable. NICE does not simply ask whether something may help an individual patient. It considers whether the available evidence justifies national funding when resources must be prioritised across the entire health service. Every funded treatment sits within that wider system.
What NICE Said About Radiotherapy for Dupuytren’s
Radiotherapy for early Dupuytren’s disease was first reviewed by NICE in 2010. The conclusion was that the treatment appeared safe, but that there was insufficient high-level evidence to support routine NHS commissioning. NICE stated that it could be offered under appropriate governance and audit arrangements.
The topic was reviewed again in 2016, with a similar outcome. The discussion focused not on safety, but on the structure of the evidence base and the absence of a placebo-controlled randomised trial.
Understanding the Evidence Debate
In modern medicine, placebo-controlled randomised trials sit at the top of the evidence hierarchy.
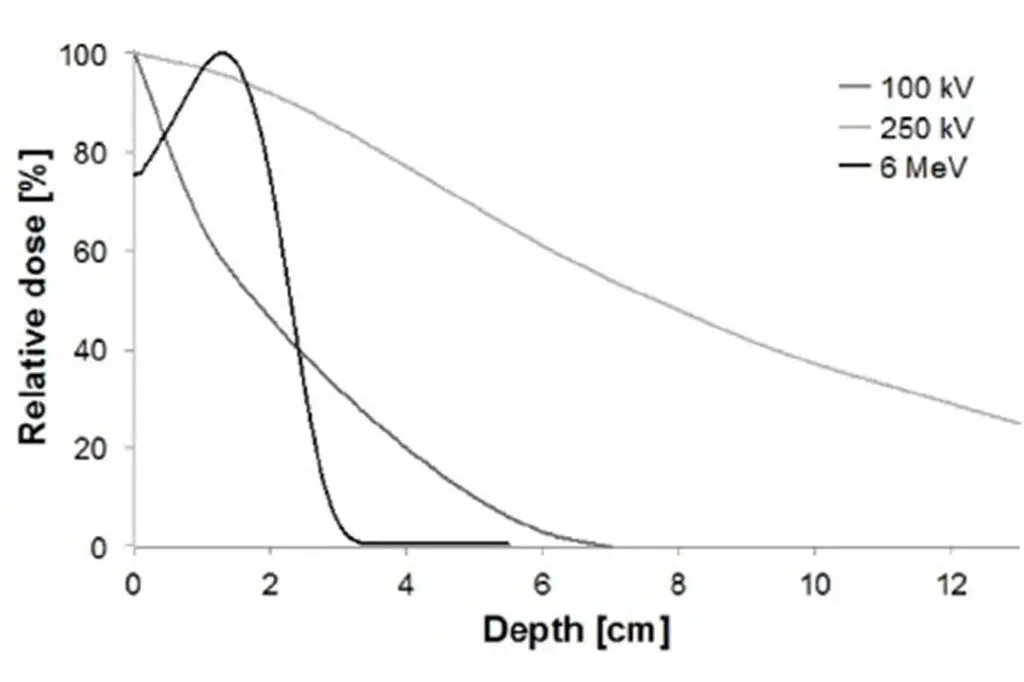
Radiotherapy for Dupuytren’s disease has been used for many years in Germany and elsewhere in Europe. Large prospective studies – including the well-known work led by Professor Seegenschmiedt – have demonstrated meaningful reductions in disease progression. In that study, patients were randomised between two radiotherapy dose levels and compared with a control group who did not receive treatment. Both radiotherapy groups showed substantially lower rates of progression than the control group, particularly the standard-dose group.
The control group consisted of patients who declined radiotherapy rather than being randomly assigned. That design has been criticised as a potential source of bias. However, the groups were carefully analysed and shown to be equivalent across known risk factors, and some baseline features were actually slightly worse in the radiotherapy group.
The design reflected clinical reality in Germany, where radiotherapy was already an established practice and patients were reluctant to be randomised to no treatment. From a NICE perspective, however, the absence of a placebo-controlled trial makes routine national commissioning more difficult to justify.
A further randomised trial – the DEPART trial in Australia – has now completed recruitment. Its results are awaited. If it confirms a substantial reduction in progression and contracture requiring surgery, that would strengthen the evidence base further and may prompt a fresh NICE appraisal, because within NHS decision-making, the structure of the evidence carries significant weight.
The Funding Context
Even low-dose radiotherapy requires resources. It involves radiotherapy machine time, radiographer time, consultant input, and treatment planning capacity. These same machines are primarily used for cancer treatment, and many NHS radiotherapy departments operate with very limited spare capacity. In some regions, cancer services themselves are under pressure, and patients may already face longer waits than ideal.
Commissioners – the organisations that pay for treatments – must decide how resources are allocated. Dupuytren’s disease is not life-threatening and often progresses slowly. When NICE guidance does not mandate funding, local commissioners may conclude that radiotherapy for Dupuytren’s does not fall within their commissioning priorities, particularly when weighed against competing demands.
Expert Non-Invasive Treatment for Dupuytren’s and Ledderhose Disease Across the UK
Dr Richard Shaffer set up Dupuytren’s UK in April 2011 to provide non-invasive treatments for people who suffer from benign conditions such as Ledderhose disease and early-stage Dupuytren’s contracture in the Guildford area. Today, Dupuytren’s UK offers this service to patients throughout the UK.
Why It Has Been Offered in Some NHS Centres
There have been NHS centres where radiotherapy for Dupuytren’s disease was offered for a period of time. Local clinical leadership and funding arrangements can sometimes allow treatments to develop outside formal national commissioning pathways.
However, funding arrangements are periodically reviewed. If commissioners decide that a treatment does not meet agreed criteria for routine funding, it may be withdrawn. Over time, access within the NHS has therefore reduced.
My Own Experience
Until 2019, I worked within the NHS and applied for radiotherapy for early Dupuytren’s disease to be funded locally. The application was declined. As a result, I began offering the treatment privately; otherwise, patients in the UK would effectively have no access to it at all.
The Current Position
At present, radiotherapy for early Dupuytren’s disease:
- Is permitted in the UK
- Is regarded as safe when delivered appropriately
- Has supportive clinical data demonstrating reduced progression
- Is not routinely commissioned by the NHS
- Is therefore usually accessed privately
For some patients, funding treatment themselves is a significant financial decision. For others – particularly those whose work, hobbies, or daily function depend heavily on their hands – the potential to reduce progression and preserve function makes it worthwhile. Where patients are covered by private medical insurance, the financial aspect may be less of a barrier, allowing the decision to focus more purely on the medical considerations.
Where I Stand
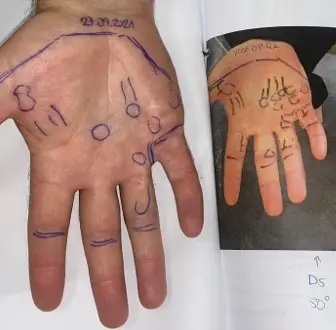
Patients with early Dupuytren’s disease are often told that nothing can be done until the fingers bend and contracture develops. Radiotherapy offers a proactive option at an earlier stage, with the aim of reducing the likelihood of contracture and preserving hand function.
I look forward to the results of the DEPART trial. If they are positive, they will add to the already substantial body of evidence supporting this treatment and may strengthen the case for wider NHS availability in the coming years.